Energy and Place
How does energy production and consumption impact place?
How does your sense of place, environmental ethic and understanding of our energy needs
influence your perception and decisions relating to energy production and consumption?
Building a Simple Household Battery
Ethan Lotfinia, Chris Niles, Sean Nickels
Animas High School
May 2015
Abstract
The motivation for this lab was to see if it is possible to build a small battery made out of materials that can be found in most households. The problem that we were trying to solve in this lab was to see if it is possible to create an efficient battery out of common household materials and chemicals. We went about working through this lab by first gathering all the materials that we thought would be required to complete it. These materials were aluminum, zinc, copper, Drano, NaCl (salt), as well as plastic tupperware and coffee stir sticks. We had followed a basic diagram of how to build the battery. The original design was layers of aluminum and copper stacked on top of each other and separated with stir sticks while connected to alligator clips. All of this was submerged in a mixture of Drano and NaCl. The changes made to the lab consisted of replacing the aluminum that was supposed to be used with zinc. We had also tried to test a version that used aluminum instead of zinc. It was observed that the zinc generated a much higher voltage than aluminum. This experiment has shown what materials would be more efficient in a battery so companies can make better products.
Introduction
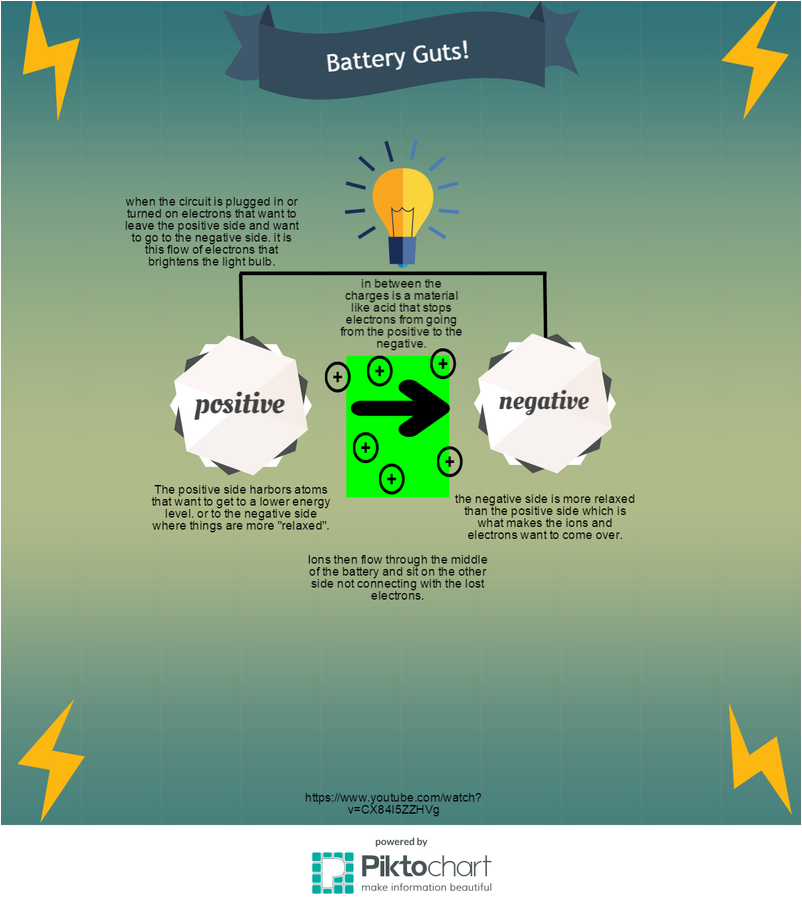
For this lab, our group decided to do an experiment in which we would build a small, simple battery using materials that could be found in most households. One reason we decided to do this lab was because our group had no prior knowledge about batteries and was curious about how we could make one. To be able to do this, we had to first gather all the materials that we would need to build the battery. A battery is able to create energy because of the electrons. The light bulb powers when electrons flow through the circuit, so knowing this the positive side of a battery holds the positively charged atoms. When the circuit is connected the electrons take this path to get to the negative side. Their spent ions move from the positive side to the negative side, however they do not connect with their lost electrons.
Methods
In this lab, drano with dissociated NaCl (table salt) was used as an electrolyte for this battery. Copper, zinc, and aluminum were used for the electrodes. The copper is used for both tests and the zinc and aluminum are used in separate tests. The metals were glued to two spacers which, in this case, were coffee stir straws. After they were glued on, alligator clips were placed on each of the metal sheets. The copper was connected with the other copper plates creating the positive charge and the zinc or aluminum was the negative charge. Both were left with an alligator clip unhooked which is where it would be hooked up to a motor. The metal plates were placed in a small container that will also house the NaCl/Drano mix. Alternate between copper and zinc or aluminum. the plastic tupperware was then filled with the drano and salt mixture. then test the battery with two different motors using a voltage meter to record the voltage and current that is produced from the reaction.
Hazards
Group members worked with stable substances throughout the entirety of the lab, with the exception of using Drano as a solution that the battery was set in. To avoid the possibility of any harm coming to group members, students had worked in a fume hood to help prevent any harmful toxins that could have been created if the drano was accidentally mixed with any other acidic substances like bleach or ammonia which would create harmful gasses. The risk of electrocution during this lab was non-existent. The voltage and current of this battery were nowhere near enough to harm any group members. Other safety precautions such as goggles and gloves were optional but not required. Getting Drano on skin was not a problem as it can be simply washed off with soap and water. Spilling Drano on clothes will result in a stain similar to that of bleach.
results
The results of this lab reflect the effectiveness of the battery we made. The zinc/copper combination was able to produce small amounts of electricity when mixed with the Drano/NaCl mixture. The initial battery was able to produce just under 1.5 volts of electricity after 10 seconds of putting the Drano mixture in the container with the metals. After 30 seconds, the voltage dropped by more than 45% from the original measurement to 0.8 volts. The aluminum/copper mixture was relatively unsuccessful due largely in part to the fact that our test was not planned enough. By the time we were trying to test the second battery with the aluminum the glue that was holding the small strips of metal togeather was striping off, this could be due to the fact that we used hot glue instead silicone sealant.
Conclusion/Discussion
The results that we had collected from this lab could be important because they could provide alternative paths for people to generate electricity in their homes. This can be used as a starting experient to families looking for alternative routes of power or also as an experiment to help someone like a student better understand how a battery works. This battery we made was not revolutionary in anyway but it can be used to teach or learn basic concepts of a battery and this could help someone design a new battery The results also show that it is possible to generate electricity from home items which is surprisingly similar to the materials they use in normal batteries.
It was expected that the aluminum/copper mixture would generate more electricity, due to results collected in a previous lab, but that was proven to be wrong after we had observed that the zinc/copper mix had created a much higher electric current than that of the aluminum. This was most likely due to the lack of understanding of what we had to do for the lab. Throughout the lab we had ran into multiple different problems that we had trouble fixing, such as how we had trouble with how our metal strips with the alligator clips sat in the tupperware. we also had problems with how small our strips of copper and zinc were we tried to glue them together but it did not seem to stick well enough for us to perform the lab correctly. If we were to redo this lab we would most likely try to get larger pieces of both copper and zinc. Another thing we would do is to also try and find a different way to have the metal strips connect with the alligator clips so they can sit evenly and correctly within our container.
How does your sense of place, environmental ethic and understanding of our energy needs
influence your perception and decisions relating to energy production and consumption?
Building a Simple Household Battery
Ethan Lotfinia, Chris Niles, Sean Nickels
Animas High School
May 2015
Abstract
The motivation for this lab was to see if it is possible to build a small battery made out of materials that can be found in most households. The problem that we were trying to solve in this lab was to see if it is possible to create an efficient battery out of common household materials and chemicals. We went about working through this lab by first gathering all the materials that we thought would be required to complete it. These materials were aluminum, zinc, copper, Drano, NaCl (salt), as well as plastic tupperware and coffee stir sticks. We had followed a basic diagram of how to build the battery. The original design was layers of aluminum and copper stacked on top of each other and separated with stir sticks while connected to alligator clips. All of this was submerged in a mixture of Drano and NaCl. The changes made to the lab consisted of replacing the aluminum that was supposed to be used with zinc. We had also tried to test a version that used aluminum instead of zinc. It was observed that the zinc generated a much higher voltage than aluminum. This experiment has shown what materials would be more efficient in a battery so companies can make better products.
Introduction
For this lab, our group decided to do an experiment in which we would build a small, simple battery using materials that could be found in most households. One reason we decided to do this lab was because our group had no prior knowledge about batteries and was curious about how we could make one. To be able to do this, we had to first gather all the materials that we would need to build the battery. A battery is able to create energy because of the electrons. The light bulb powers when electrons flow through the circuit, so knowing this the positive side of a battery holds the positively charged atoms. When the circuit is connected the electrons take this path to get to the negative side. Their spent ions move from the positive side to the negative side, however they do not connect with their lost electrons.
Methods
In this lab, drano with dissociated NaCl (table salt) was used as an electrolyte for this battery. Copper, zinc, and aluminum were used for the electrodes. The copper is used for both tests and the zinc and aluminum are used in separate tests. The metals were glued to two spacers which, in this case, were coffee stir straws. After they were glued on, alligator clips were placed on each of the metal sheets. The copper was connected with the other copper plates creating the positive charge and the zinc or aluminum was the negative charge. Both were left with an alligator clip unhooked which is where it would be hooked up to a motor. The metal plates were placed in a small container that will also house the NaCl/Drano mix. Alternate between copper and zinc or aluminum. the plastic tupperware was then filled with the drano and salt mixture. then test the battery with two different motors using a voltage meter to record the voltage and current that is produced from the reaction.
Hazards
Group members worked with stable substances throughout the entirety of the lab, with the exception of using Drano as a solution that the battery was set in. To avoid the possibility of any harm coming to group members, students had worked in a fume hood to help prevent any harmful toxins that could have been created if the drano was accidentally mixed with any other acidic substances like bleach or ammonia which would create harmful gasses. The risk of electrocution during this lab was non-existent. The voltage and current of this battery were nowhere near enough to harm any group members. Other safety precautions such as goggles and gloves were optional but not required. Getting Drano on skin was not a problem as it can be simply washed off with soap and water. Spilling Drano on clothes will result in a stain similar to that of bleach.
results
The results of this lab reflect the effectiveness of the battery we made. The zinc/copper combination was able to produce small amounts of electricity when mixed with the Drano/NaCl mixture. The initial battery was able to produce just under 1.5 volts of electricity after 10 seconds of putting the Drano mixture in the container with the metals. After 30 seconds, the voltage dropped by more than 45% from the original measurement to 0.8 volts. The aluminum/copper mixture was relatively unsuccessful due largely in part to the fact that our test was not planned enough. By the time we were trying to test the second battery with the aluminum the glue that was holding the small strips of metal togeather was striping off, this could be due to the fact that we used hot glue instead silicone sealant.
Conclusion/Discussion
The results that we had collected from this lab could be important because they could provide alternative paths for people to generate electricity in their homes. This can be used as a starting experient to families looking for alternative routes of power or also as an experiment to help someone like a student better understand how a battery works. This battery we made was not revolutionary in anyway but it can be used to teach or learn basic concepts of a battery and this could help someone design a new battery The results also show that it is possible to generate electricity from home items which is surprisingly similar to the materials they use in normal batteries.
It was expected that the aluminum/copper mixture would generate more electricity, due to results collected in a previous lab, but that was proven to be wrong after we had observed that the zinc/copper mix had created a much higher electric current than that of the aluminum. This was most likely due to the lack of understanding of what we had to do for the lab. Throughout the lab we had ran into multiple different problems that we had trouble fixing, such as how we had trouble with how our metal strips with the alligator clips sat in the tupperware. we also had problems with how small our strips of copper and zinc were we tried to glue them together but it did not seem to stick well enough for us to perform the lab correctly. If we were to redo this lab we would most likely try to get larger pieces of both copper and zinc. Another thing we would do is to also try and find a different way to have the metal strips connect with the alligator clips so they can sit evenly and correctly within our container.
Reflection
Our lab was making a battery out of household material. This lab was very interesting and started to put in perspective how a battery works but also the waste that may come from a battery. When we assembled the battery we used different materials, from acids to metals to even plastics. If these kinds of thing are out of household batteries I would imagine that normal batteries use a stronger acid than Drano. Although batteries provide energy, the waste that disposable batteries create seems like too much pollution.
I decided to make my infographic about how a simple rechargeable battery works. To do this, I did a little more research to figure out how a battery works. I try to explain the positive and negative sides of a battery, along with explaining the acidic part inside of the battery. I wanted to explain the reverse side of the battery which would be the recharging part but sadly I did not have the time to add more detail.
Our lab was making a battery out of household material. This lab was very interesting and started to put in perspective how a battery works but also the waste that may come from a battery. When we assembled the battery we used different materials, from acids to metals to even plastics. If these kinds of thing are out of household batteries I would imagine that normal batteries use a stronger acid than Drano. Although batteries provide energy, the waste that disposable batteries create seems like too much pollution.
I decided to make my infographic about how a simple rechargeable battery works. To do this, I did a little more research to figure out how a battery works. I try to explain the positive and negative sides of a battery, along with explaining the acidic part inside of the battery. I wanted to explain the reverse side of the battery which would be the recharging part but sadly I did not have the time to add more detail.
Elevator Pitch
Ohh no not my phone! What a tragedy! Hi Sean here, have you ever known someone or even had a broken phone screen? Well for my project I have looked in to fixing that problem! Current phones use gorilla glass which is a glass that is always chemically stressed so the impact has to overcome that stress to break the screen, the way that it is stressed is by replacing a smaller atom in the glass with a bigger one so it pushes up against the others. Although once this glass breaks it shatters badly making spider web fractures all throughout the phone. My proposal was the glass have a layer of thermoly conductive plastic underneath the glass so when there is an impact it is dampened by the plastic which is able to take shock easier! The plastic would be made of a carbon material because it is found that that is not only very strong but also conductive to allow the touch screen to work. The glass is placed on top because it is a harder surface and unlike the plastic it will not scratch from keys or other random items in ones pocket. This is kind of like the idea for car windows or even bullet proof glass. Now it wouldn’t be indestructible but it would allow the glass to be more forgiving to higher droops thus protecting your screen. Now I would like money to fund this idea and you get to stop buying new phone screens or even new phones!
letter proposal
Chemistry has changed and shaped how we live today. so many things have been changed as a result of chemistry, for example with the recent technology we have been able to form a stronger type of glass that has been used recently as phone screens. This has been an achievement in chemistry because of the process used to hardened the glass is that a molecule is replaced with a different larger molecule thus keeping the glass tight and under stress.Even with this I think it is very possible to take this even further and develop a better phone screen that can handle better with out being broken. That is why I choose to write my project on a better phone screen that involves a polymer and a glass.
Atoms and molecules have many different properties when you look closely. these different properties can change the properties of a larger object. Such as I had done some research on conductive polymer, it turns out that the way the plastic is conductive is because of the chain of carbon is not fully connected to the other carbon atoms which allows an atom to move and respond thus making it conductive. This was very interesting because normally polymers are not conductive. As I explained before the gorilla glass part has also been through a chemical change putting it under more stress because of how tight the particles are compacted. This research took me quite a while to find due to the way that phone companies don't really describe how they make their screens.
Atoms and molecules have many different properties when you look closely. these different properties can change the properties of a larger object. Such as I had done some research on conductive polymer, it turns out that the way the plastic is conductive is because of the chain of carbon is not fully connected to the other carbon atoms which allows an atom to move and respond thus making it conductive. This was very interesting because normally polymers are not conductive. As I explained before the gorilla glass part has also been through a chemical change putting it under more stress because of how tight the particles are compacted. This research took me quite a while to find due to the way that phone companies don't really describe how they make their screens.